Photon Energy Calculator
Photon Energy · Wavelength · Frequency
ephotonics
linkedin.com/company/ephotonics
Follow our page for weekly laser engineering guides, photonics tutorials, and new calculator releases directly in your feed.
Follow on LinkedInHow the Photon Energy Calculator Works
Light is fundamentally made up of individual packets of energy called photons. According to the Planck-Einstein relation, the energy of a single photon is directly proportional to its frequency and inversely proportional to its wavelength.
Physical Constants Used
- E Photon Energy: The resulting energy of the light particle, measured in Joules (J).
- h Planck's Constant: A fundamental universal constant, exactly 6.62607015 × 10-34 J·s.
- c Speed of Light: The speed at which photons travel in a vacuum, exactly 299,792,458 m/s.
- λ (Lambda) Wavelength: The physical distance between wave peaks.
- f Frequency: The number of wave cycles per second.
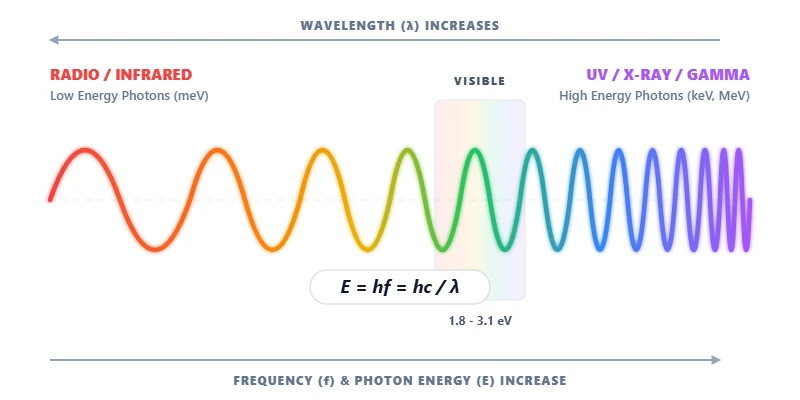
The Electron-Volt (eV) Conversion
Because the energy of a single photon measured in Joules is unfathomably small (usually on the order of 10-19 Joules), physicists and photonics engineers almost exclusively use Electron-Volts (eV) instead. One electron-volt is defined as the amount of kinetic energy gained by a single electron accelerating from rest through an electric potential difference of one volt.
Quick Rule of Thumb: Visible light photons carry energies ranging roughly from 1.8 eV (Red light) to 3.1 eV (Violet light). If your calculated energy is far outside this range, you are likely looking at the infrared or ultraviolet spectrum.
Understanding Photon Energy in Quantum Physics
In classical physics light was historically treated as a continuous electromagnetic wave. However the discovery of quantum mechanics revealed that light actually behaves as individual packets of quantized energy known as photons.
This dual nature of light means that the energy contained within a single photon is directly tied to its frequency and wavelength. By calculating this energy engineers and physicists can determine exactly how a specific beam of light will interact with different materials and sensors.
The Relationship Between Color and Energy
The color of light we see is just a physical manifestation of its energy. Red light has a longer wavelength and lower frequency meaning its photons carry less energy. Blue and violet light have much shorter wavelengths meaning their photons pack significantly more energy.
When you move beyond the visible spectrum this energy difference becomes critical. Ultraviolet light x rays and gamma rays carry so much photon energy that they become ionizing radiation capable of knocking electrons out of atoms and breaking chemical bonds. Conversely lower energy photons like infrared and microwaves only carry enough energy to cause molecular vibrations which we feel as heat.
Key Applications for Photon Energy
1. Semiconductor Bandgap Engineering
For a light emitting diode or laser diode to function an electron must drop across a specific bandgap. This drop releases a photon. Engineers use a photon energy calculator to match the material bandgap to the desired output color.
2. Photovoltaics and Solar Efficiency
Solar panels rely on the photoelectric effect. A solar cell can only convert photons that carry enough energy to knock an electron loose from the silicon lattice. Photons with energy lower than the bandgap pass through generating heat instead of power.
3. Photochemistry and Curing
Chemical reactions such as ultraviolet curing of resins or biological imaging require a minimum activation energy to proceed. By converting the activation energy into wavelength chemists can determine exactly which light source will trigger the reaction.
4. High Energy Physics
As the electromagnetic spectrum moves into x rays and gamma rays describing light by wavelength becomes cumbersome. In these high energy fields scientists abandon wavelength entirely and use this calculator to describe photons purely by their energy.
Frequently Asked Questions
How do you calculate the energy of a single photon
What is the relationship between wavelength and photon energy
Why is photon energy measured in electron volts instead of joules
Which color of visible light has the highest photon energy
How does photon energy relate to the electromagnetic spectrum
What exactly is a Photon?
Before calculating its energy, explore the fascinating physics behind the fundamental particle of light. Learn about wave-particle duality, zero rest mass, and how photons power modern technology.
Read the full guide