Laser Heat Transfer Calculator
Thermal Resistance · Heat Dissipation · Junction Temperature
ephotonics
linkedin.com/company/ephotonics
Follow our page for weekly laser engineering guides, photonics tutorials, and new calculator releases directly in your feed.
Follow on LinkedInHow the Laser Heat Transfer Calculator Works
Laser heat transfer calculator estimates instantaneous temperature rise using the principles of Adiabatic Calorimetry. It calculates the upper-limit thermal load by assuming that all absorbed laser energy is trapped entirely within the illuminated volume of the target material, prior to thermal conduction pulling the heat away.
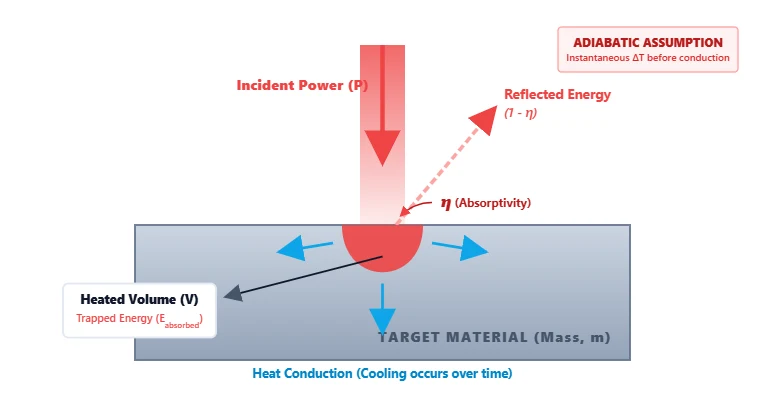
Understanding the Variables:
- Eabsorbed Absorbed Energy (J): The total laser energy ($P \times t \times \eta$) that successfully enters the material, minus reflected light.
- m Heated Mass (g): The mass of the material directly exposed to the beam, calculated as $\text{Density} (\rho) \times \text{Volume}$.
- cp Specific Heat (J/g·°C): The amount of energy required to raise the temperature of 1 gram of the material by 1 degree Celsius.
- η Absorptivity: The fraction of incident laser light that is absorbed by the material rather than reflected (e.g., 0.1 for 10% absorption).
Why Calculate Laser Heat Transfer?
- Laser Cutting/Welding: Determine laser power for precise material ablation.
- Additive Mfg.: Optimize laser scan speed for sintering powders.
- Medical Lasers: Control thermal effects for surgery (coagulation vs. ablation).
- Optics Lifetime: Prevent damage to lenses/mirrors from excessive absorption.
Understanding Laser Absorption
The key to laser material processing is how much energy the target absorbs. A highly reflective metal like Copper or Aluminum needs a very different laser setup compared to a highly absorbent material like a semiconductor or black plastic.
The Absorptivity (η) dictates how much of the incident laser power (P) actually contributes to heating. This absorbed energy (Q = P × t × η) is then distributed throughout the heated volume based on the material's Density (ρ) and Specific Heat (cp).
1. Machining Precision
To achieve a clean laser cut vs. a molten blob, you need to exceed the material's vaporization point within the laser's spot size. Laser heat transfer calculator helps determine if your parameters are correct for high-aspect ratio features.
2. Thermal Management
Even a small absorption percentage (e.g., 0.1%) can drastically increase optic temperatures. Knowing the expected heat load is essential for designing cooling systems or selecting optics with sufficient damage thresholds.
3. Medical Ablation
In laser eye surgery (LASIK) or skin resurfacing, the goal is precise material removal via rapid vaporization. This requires delivering enough energy density to reach the boiling point without causing collateral thermal damage.
4. 3D Printing (Additive Mfg)
Selective Laser Melting (SLM) or Sintering (SLS) requires careful control. You need enough energy to fuse the powder, but not so much that it vaporizes or causes excessive thermal stress and warping.
Thermal Lensing Note
When a high-power laser passes through a lens, even 0.1% absorption heats the glass. This slight temperature rise changes the refractive index (dn/dT) and expands the lens, creating a "Thermal Lens" that shifts the focus and degrades beam quality (M²).
