Every time you see a rainbow, tune a laser, or stream data through a fiber-optic cable, one number quietly governs everything: wavelength. It determines the color your eye perceives, the distance a laser beam can travel without spreading, and whether two light waves cancel each other out or combine into a blinding peak. Understanding wavelength isn’t just textbook physics, it is the foundation on which all of modern photonics is built.
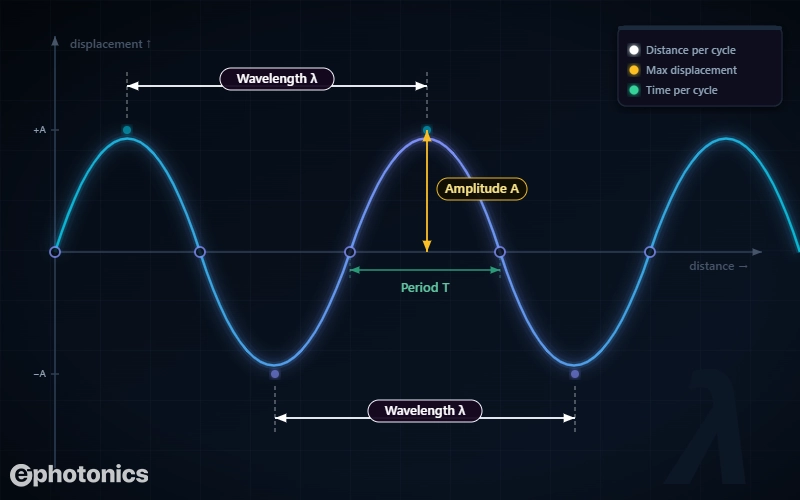
Imagine freezing a wave in time and taking a photograph. The wavelength, symbol λ (Greek letter lambda) is simply the distance from one crest to the next, or equivalently from one trough to the next, or between any two identical points one full cycle apart.
The diagram below shows three quantities at once. These three are independent of each other: you can have a high-amplitude, short-wavelength wave just as easily as a low-amplitude, long-wavelength one.
Measured horizontally. It is the physical distance between two matching points on the wave (crest to crest).
Measured vertically. It is the reach from the center-line to the crest, representing the wave’s signal strength.
Measured in time. It is the duration (in seconds) it takes for one full oscillation cycle to complete.
More formally, for the simplest possible wave, a monochromatic plane wave, the wave amplitude (A) at position r and time t is:
Variable Reference
In plain terms: travel one wavelength forward along the beam and the wave looks exactly as it did before, you’ve completed one full spatial cycle, accumulating a phase of 2π.
Quick intuition: Wavelength is to space what period is to time. Period T tells you how long one oscillation takes; wavelength tells you how much space it occupies.
Wavelength and frequency are two faces of the same coin. As a wave travels one full wavelength in space during exactly one period in time, its phase velocity vph is:
In vacuum, light always travels at c = 299,792,458 m/s (exactly, it is a defined constant in the SI system). So:
This inverse relationship is critically important: higher frequency = shorter wavelength. A violet photon at 400 nm oscillates nearly twice as fast as a red photon at 700 nm. Double the frequency, halve the wavelength, the wave just gets physically “compressed” in space. The illustration below visualizes this core concept. As light shifts from red to violet, the wavelength physically shrinks (Short λ). Because the wave is shorter, more cycles pass by every second, resulting in a higher frequency (f) and higher energy (E).
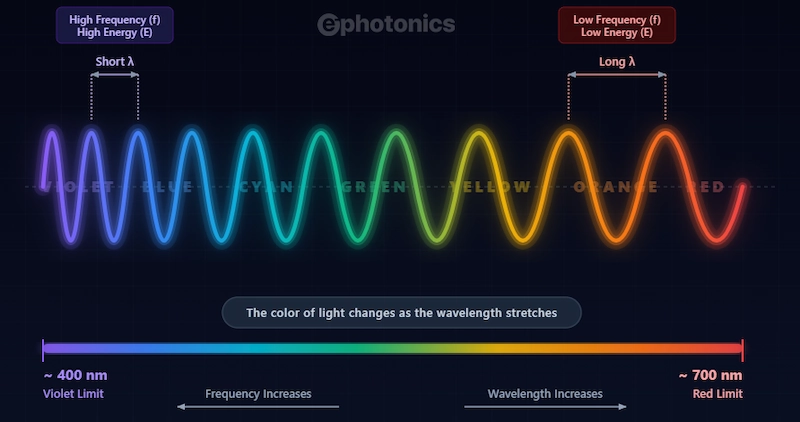
For a precise breakdown of these values, use the reference table below to see the exact vacuum wavelengths, frequencies, and photon energies for each major band of light:
| Color / Band | Vacuum λ | Frequency (f) | Photon Energy (E) |
|---|---|---|---|
| Violet | ~ 380–450 nm | ~ 665–789 THz | ~ 2.75–3.26 eV |
| Blue | ~ 450–495 nm | ~ 606–665 THz | ~ 2.50–2.75 eV |
| Green | ~ 495–570 nm | ~ 526–606 THz | ~ 2.17–2.50 eV |
| Yellow | ~ 570–590 nm | ~ 508–526 THz | ~ 2.10–2.17 eV |
| Orange | ~ 590–625 nm | ~ 480–508 THz | ~ 1.98–2.10 eV |
| Red | ~ 625–750 nm | ~ 400–480 THz | ~ 1.65–1.98 eV |
| Near-infrared | ~ 750–1400 nm | ~ 214–400 THz | ~ 0.88–1.65 eV |
| Telecom C-band | ~ 1530–1565 nm | ~ 192–196 THz | ~ 0.79–0.81 eV |
Not all color comes from pigment. While a red apple absorbs every wavelength except red, some of the most vibrant colors in nature, like the wings of a Morpho butterfly or the feathers of a peacock are created by geometry, not chemistry. This is called Structural Coloration.
These organisms have microscopic nanostructures on their surface that are perfectly spaced to match specific wavelengths of light. When white sunlight hits these structures, only specific wavelengths (usually bright blues and greens) undergo constructive interference and reflect back to your eye, while other wavelengths interfere destructively and vanish.
This is exactly how a Fiber Bragg Grating works in the telecom industry. We didn’t invent interference filters; nature did, millions of years ago. By adjusting the spacing of these biological nanostructures, nature effectively “tunes” the wavelength of the animal, creating iridescent colors that never fade because they rely on physics, not dye.
Light visible to the human eye occupies only a narrow sliver, roughly 380 nm to 750 nm of a spectrum that stretches from kilometre-long radio waves down to sub-picometer gamma rays.
To put this massive scale into perspective, try out the Electromagnetic Spectrum Simulator below. Drag the white node along the wave to travel through the different frequency bands. As the wavelength compresses, keep an eye on the Comparable Size indicator in the bottom corner to see how these invisible waves measure up to real-world objects from buildings all the way down to atomic nuclei.
 ElectroMAGNETIC Spectrum Simulator
ElectroMAGNETIC Spectrum Simulator
While the simulation above shows the vastness of the electromagnetic spectrum, the optics industry relies on highly specific bandwidths. Because different wavelengths interact with matter in unique ways, they are harnessed for very different technologies. Here is a breakdown of the most commercially important photonics bands and their primary applications:
| Spectrum Band | Wavelength Range | Commercial Applications |
|---|---|---|
| Ultraviolet (UV) | 200–380 nm | Semiconductor lithography Sterilisation Fluorescence microscopy |
| Visible Light | 380–750 nm | Displays Ophthalmology Bio-imaging RGB lasers |
| Near-Infrared (NIR) | 750–1400 nm | Silicon photonics Laser surgery LiDAR |
| Telecom Bands | 1260–1625 nm | Fiber-optic communications Dense WDM |
| Mid-Infrared (MIR) | 2–20 µm | Molecular fingerprinting Thermal imaging Gas sensing |
Here is a subtlety that trips up even experienced researchers. When light enters a material such as glass, water, a semiconductor, its frequency stays constant, but its speed drops by a factor equal to the refractive index n:
Since λ = v/ν, the λ inside the medium shrinks proportionally:
To see exactly what these equations mean in the physical world, look at the boundary diagram below. Notice how the wave bunches up the moment it hits the denser glass. Because the light is forced to slow down, the physical distance between each wave peak has to compress to keep the frequency perfectly constant.
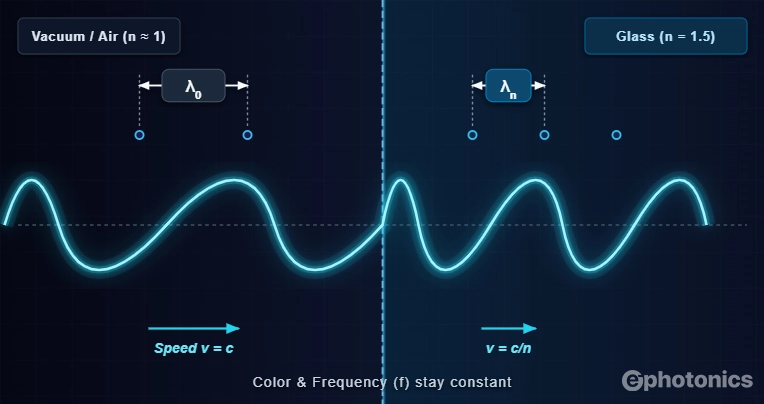
For example, a 1064 nm Nd:YAG laser beam inside a glass lens with n = 1.5 has an in-medium λ of only 709 nm, even though we still call it 1064 nm light. That number refers to its vacuum wavelength, which is the universally agreed standard for identifying light.
Why vacuum wavelength? Because the refractive index of air itself varies with pressure, temperature, humidity, and CO₂ concentration. Standard dry air at 15 °C and 1013.25 mbar shifts a 1000 nm vacuum λ to ~999.73 nm in air, a 0.027% difference that is negligible for most purposes but matters enormously in precision interferometry and laser spectroscopy.
Always report vacuum wavelengths in your publications. It completely removes ambiguity and allows anyone to accurately compute the in-medium wavelength, provided they know the refractive index.
Wavelength is not just a label we use to name a color. It is a fundamental physical parameter that actively governs how light behaves in every optical system. Whether you are designing a microscope, a fiber-optic network, or a lithography stepper, the wavelength (λ) dictates the ultimate performance limits of your device. Here are the four critical physical mechanisms where wavelength plays the leading role.
The longer the wavelength relative to an aperture, the more a beam spreads. This is why 1550 nm light diffracts more than 532 nm. The Rayleigh range scales with λ, which is why chip manufacturers use 13.5 nm EUV light to print sub-10nm features.
Constructive or destructive interference depends entirely on the optical path difference relative to λ. A difference of just λ/2 flips the signal from maximum to zero.
The Rayleigh criterion sets the ultimate limit of any optical system. Resolution improves linearly as wavelength decreases. This is why astronomers build larger mirrors and biologists use UV microscopy.
In nonlinear optics (like second-harmonic generation), efficiency relies on maintaining a fixed phase relationship. Because refractive index changes with wavelength, engineering the exact λ is the central challenge in nonlinear photonics.
In the laboratory, calculating v=c/n isn't enough. We need to measure real laser sources with extreme accuracy. Here are the three industry-standard instruments used to quantify light, ranked by precision.
OSAs disperse light via diffraction gratings to record power at each λ. They are the workhorse for visualizing the shape of a spectrum. They report units of W/nm or dBm/nm, allowing you to read peak λ, bandwidth, and side-mode suppression ratio (SMSR) directly.
A wavemeter is a specialized interferometer that compares the fringe spacing of an unknown beam against a known reference source (typically a stabilised HeNe laser). While they cannot visualize the spectrum shape like an OSA, they achieve significantly higher absolute accuracy — up to sub-femtometre precision in top-tier laboratory instruments.
This is the ultimate limit of measurement. An optical frequency comb emits a precise "comb" of millions of sharp frequency lines, enabling absolute frequency measurement at the 10-18 level. The vacuum λ is derived with perfect accuracy using λ0 = c / ν.
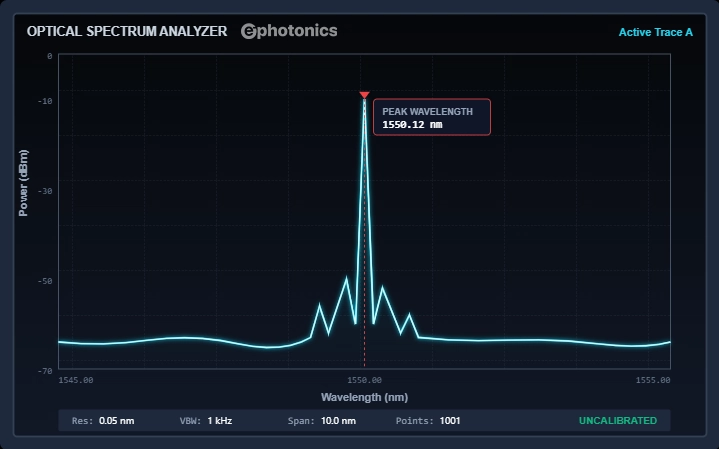
Real light sources such as LEDs, ultrashort laser pulses, thermal sources are never perfectly monochromatic. They span a range of wavelengths described by their optical spectrum. Two quantities then matter:
For a femtosecond pulse with a bandwidth of 10 nm around 800 nm, the centre and peak λ may differ by only a fraction of a nanometre but that difference is measurable and matters in ultrafast experiments.
Often, you need to convert between λ bandwidth (Δλ) and frequency bandwidth (Δν). This relationship relies on the derivative of the wave equation:
Don't calculate Δν manually. Instantly switch between Wavelength and Frequency.
In practical optical engineering, λ is rarely just a number, it dictates the physical geometry and material properties of every component you build. Whether you are cleaving a fiber or temperature-tuning a laser, the operating wavelength drives the fundamental design rules. Here is a quick reference guide to how wavelength governs the most critical devices in a photonics lab:
| Device | Key Wavelength Concept |
|---|---|
|
Fiber Bragg Grating
|
Bragg wavelength (peak reflectivity): λB = 2nΛ |
|
Single-mode fiber
|
Cut-off λ: The longest λ at which higher-order modes are still guided. Above this, it is strictly single-mode. |
|
DFB Laser
|
Emission wavelength is strictly set by the internal grating period. |
|
WDM System
|
Channel spacing defined in nm or GHz on the standard ITU grid. |
|
AR Coating
|
Layer thickness must be exactly quarter-wave optical thickness (λ/4) at the design wavelength. |
|
Nonlinear Crystal (SHG)
|
Output at λ/2 via phase-matched second-harmonic generation. |
|
Photodetector
|
Cut-off wavelength set by bandgap energy: λc = hc / Eg |
Wavelength is the fundamental ruler of the optical world. From the 13.5 nm EUV light printing our processors to the 1550 nm signals carrying the internet, understanding exact wavelengths allows engineers to control light with precision. Whether you are aligning a laser or analyzing a spectrum, remember: frequency is the identity, but wavelength is the behavior.
Join our newsletter to stay updated with new articles and news!

ephotonics is a go-to partner for photonics solutions, with deep expertise in laser electronics and laser design. We’re committed to delivering effective solutions for laser-based applications and sharing helpful articles. To learn more about us, check out our About page.