Just think about a sunny day outside, where the heat of the sun touches your skin. Actually, you are being bombarded by billions of tiny particles called photons streaming from the Sun across 93 million miles of space to reach Earth. Moving with such energy, these particles light up all that they touch, heating your skin up at the speed of light. Amazingly, the heat and light you feel are supplied by interactions at the microscopic level: photons interacting with and energizing the molecules of your skin that sense temperature.
But what exactly are these photons? They are not just particles of light; they are something more. They are fundamental building blocks of the universe that, in fact, help form an in-depth understanding of energy and matter, not to mention the very nature of light. They lie at the very core of that catchy little concept called wave-particle duality, wherein the photons behave as both particles and waves.
A photon is a massless, elementary particle that is the fundamental unit of light and all other forms of electromagnetic radiation. Photons have neither rest mass nor electric charge but they do have energy and momentum, which enables them to exert a force. They always travel at the speed of light (about 299,792 kilometres per second in a vacuum) and their energies are proportional to, but independent of power so, their frequency expressed as:
E = hν
where E is the energy of the photon, h is the Planck’s constant and ν is the frequency of the light wave. If you would like to calculate energy of a photon from its wavelength or frequency, you could use our photon energy calculator.
To truly understand what a photon is, we have to look at how it is born. They don’t just appear out of nowhere; they are the direct result of atomic interactionsi specifically, shifting the electrons. Imagine an atom as a miniature solar system, where electrons orbit a central nucleus at very specific, fixed distances called energy levels. When an atom absorbs energy (like heat or electricity), its electrons become “excited” and jump outward to a higher, unstable energy orbit.
But electrons want to be stable. Almost instantly, they drop back down to their original, lower-energy orbit. Because energy cannot simply disappear, the exact amount of energy the electron loses during this drop is expelled outward into space. This expelled packet of energy is a photon.
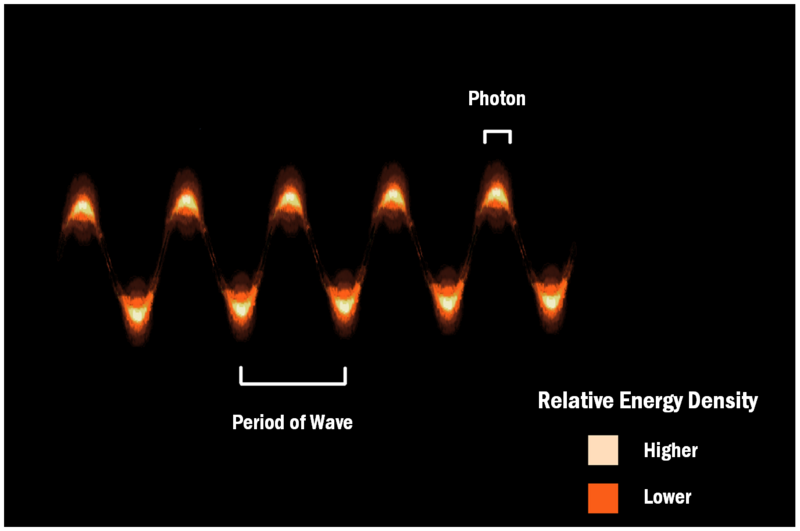
The energy of this newly born photon (E) is directly tied to the frequency (ν) and wavelength (λ) of its wave, a relationship beautifully summarized by the diagram below.
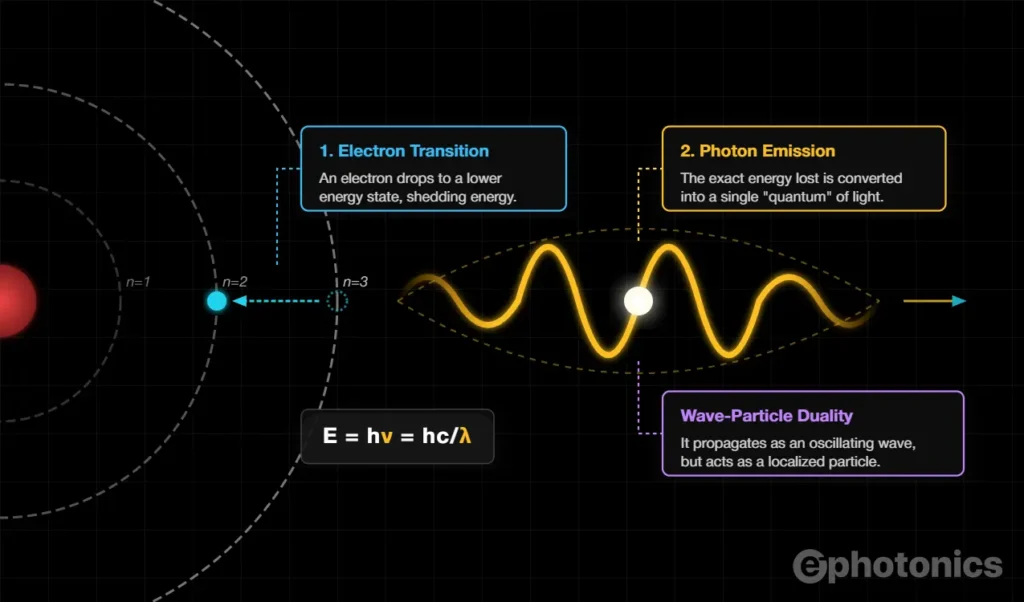
One of the interesting things relating to photons is that they sit at an intersection between waves and particles-a principle known as wave-particle duality. In their wave properties, they have wavelength and frequency, which will define what kind of electromagnetic radiation they represent. The only differences between photons in visible light and those in X-rays or radio waves are frequency and wavelength.
Yet, they also behave as if they are particles, mainly when they interact with matter. Indeed, this was spectacularly confirmed in 1905 when Albert Einstein explained such a thing as the photoelectric effect in his paper titled “On a Heuristic Point of View Concerning the Production and Transformation of Light” He showed that light delivers its energy in discrete packets, or “quanta,” now what we call photons. That the discovery of light had particle properties won Einstein a Nobel Prize and set the stage for quantum mechanics.
Basically, they are important because they are the major carrier of energy and information across the universe. As the basic units of light and all other electromagnetic radiations, they make vision possible and drive photosynthesis, hence maintaining life on Earth. They lie at the heart of state-of-the-art technologies, serving, for example, in telecommunications for the transmission of data at high speed using optical fibers, or in medical imagery for the visualization of the interior of the body through techniques such as X-rays and PET scans.
The same could be said of renewable energies, where solar panels transform photons into electrical energy thanks to the photoelectric effect. Beyond practical applications, they are are also driving research in quantum mechanics and cosmology, where their properties may yield insights into the structure of matter, time, and space itself. Therefore, photons light up not just our physical world but also our knowledge of basic sciences and technologies.
Join our newsletter to stay updated with new articles and news!

ephotonics is a go-to partner for photonics solutions, with deep expertise in laser electronics and laser design. We’re committed to delivering effective solutions for laser-based applications and sharing helpful articles. To learn more about us, check out our About page.